Amendment Chart
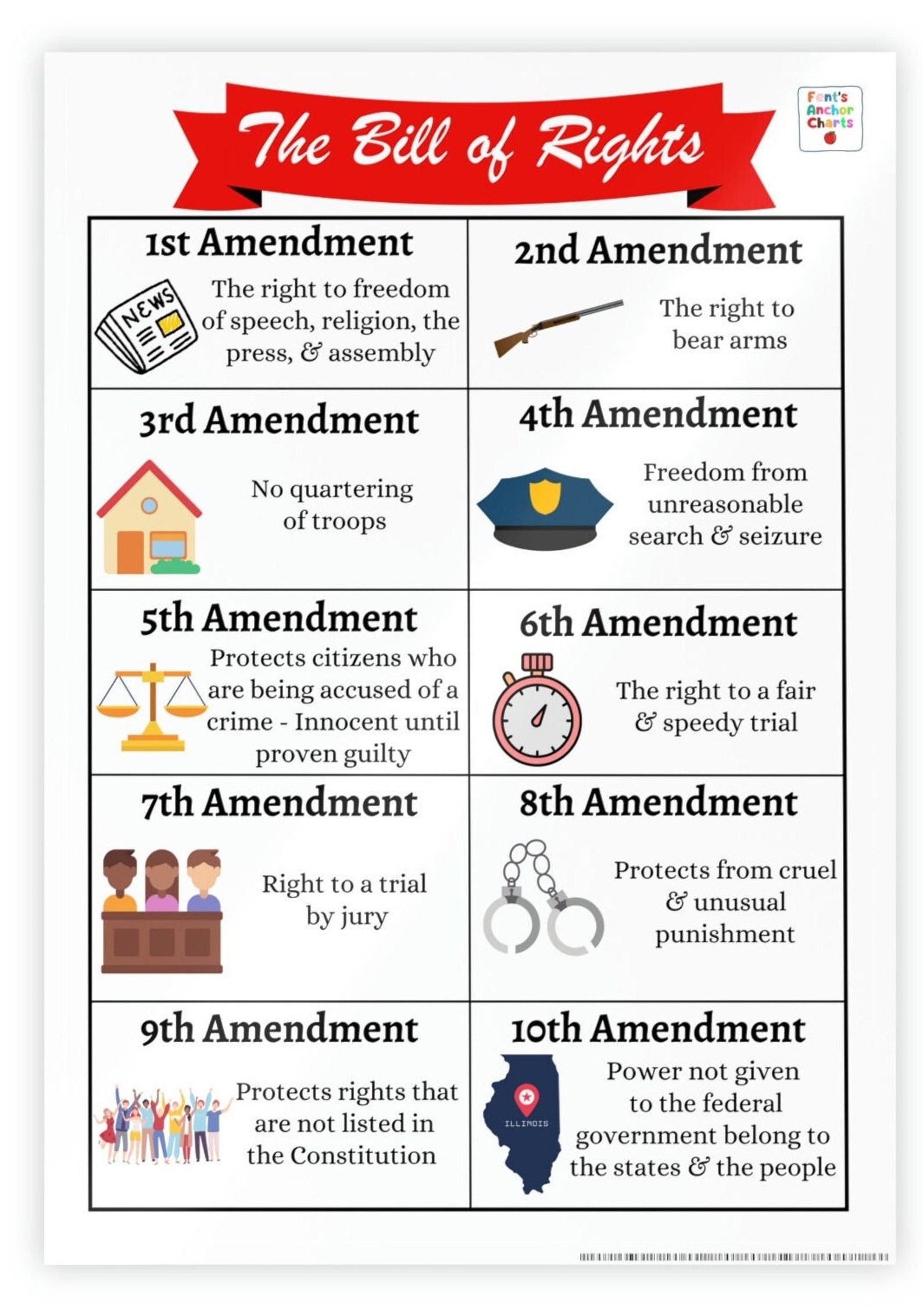
Amendment Chart - Amendments may be submitted at anytime, however, the change must not be. The irb reviews protocols to ensure appropriate safeguards to protect the rights and welfare of research subjects are in place, according to 45 cfr 46.111. Overview you can only request modifications if your study is active and approved by the irb. Can i submit an amendment to my current irb? The information provided here builds on. To make changes to your approved study, please follow the instructions below. Investigators with an approved protocol must submit an amendment/modification of research form if there are significant changes involving any of the study protocols, study design,. The guidance put forth by byu human research protection program borrows extensively from the best practices of a variety of academic institutions. Yes, if your study is currently approved through the pdf forms, you can continue to submit amendments on the pdf forms until you convert. Before changes can be made to an approved irb protocol, an amendment must be submitted to the irb. Can i submit an amendment to my current irb? Before changes can be made to an approved irb protocol, an amendment must be submitted to the irb. The guidance put forth by byu human research protection program borrows extensively from the best practices of a variety of academic institutions. Overview you can only request modifications if your study is active and approved by the irb. The information provided here builds on. Investigators with an approved protocol must submit an amendment/modification of research form if there are significant changes involving any of the study protocols, study design,. The irb reviews protocols to ensure appropriate safeguards to protect the rights and welfare of research subjects are in place, according to 45 cfr 46.111. To make changes to your approved study, please follow the instructions below. Yes, if your study is currently approved through the pdf forms, you can continue to submit amendments on the pdf forms until you convert. Amendments may be submitted at anytime, however, the change must not be. To make changes to your approved study, please follow the instructions below. Yes, if your study is currently approved through the pdf forms, you can continue to submit amendments on the pdf forms until you convert. Amendments may be submitted at anytime, however, the change must not be. Can i submit an amendment to my current irb? The irb reviews. Amendments may be submitted at anytime, however, the change must not be. Can i submit an amendment to my current irb? To make changes to your approved study, please follow the instructions below. Investigators with an approved protocol must submit an amendment/modification of research form if there are significant changes involving any of the study protocols, study design,. Yes, if. To make changes to your approved study, please follow the instructions below. The guidance put forth by byu human research protection program borrows extensively from the best practices of a variety of academic institutions. Investigators with an approved protocol must submit an amendment/modification of research form if there are significant changes involving any of the study protocols, study design,. The. Before changes can be made to an approved irb protocol, an amendment must be submitted to the irb. The information provided here builds on. To make changes to your approved study, please follow the instructions below. Can i submit an amendment to my current irb? The irb reviews protocols to ensure appropriate safeguards to protect the rights and welfare of. Overview you can only request modifications if your study is active and approved by the irb. Can i submit an amendment to my current irb? The guidance put forth by byu human research protection program borrows extensively from the best practices of a variety of academic institutions. Investigators with an approved protocol must submit an amendment/modification of research form if. Amendments may be submitted at anytime, however, the change must not be. Can i submit an amendment to my current irb? The irb reviews protocols to ensure appropriate safeguards to protect the rights and welfare of research subjects are in place, according to 45 cfr 46.111. The guidance put forth by byu human research protection program borrows extensively from the. Can i submit an amendment to my current irb? The information provided here builds on. To make changes to your approved study, please follow the instructions below. Before changes can be made to an approved irb protocol, an amendment must be submitted to the irb. Yes, if your study is currently approved through the pdf forms, you can continue to. Can i submit an amendment to my current irb? The guidance put forth by byu human research protection program borrows extensively from the best practices of a variety of academic institutions. The irb reviews protocols to ensure appropriate safeguards to protect the rights and welfare of research subjects are in place, according to 45 cfr 46.111. The information provided here. Yes, if your study is currently approved through the pdf forms, you can continue to submit amendments on the pdf forms until you convert. The guidance put forth by byu human research protection program borrows extensively from the best practices of a variety of academic institutions. To make changes to your approved study, please follow the instructions below. The information. Can i submit an amendment to my current irb? Before changes can be made to an approved irb protocol, an amendment must be submitted to the irb. Yes, if your study is currently approved through the pdf forms, you can continue to submit amendments on the pdf forms until you convert. Overview you can only request modifications if your study. Overview you can only request modifications if your study is active and approved by the irb. Can i submit an amendment to my current irb? Before changes can be made to an approved irb protocol, an amendment must be submitted to the irb. The irb reviews protocols to ensure appropriate safeguards to protect the rights and welfare of research subjects are in place, according to 45 cfr 46.111. The guidance put forth by byu human research protection program borrows extensively from the best practices of a variety of academic institutions. Yes, if your study is currently approved through the pdf forms, you can continue to submit amendments on the pdf forms until you convert. To make changes to your approved study, please follow the instructions below. Amendments may be submitted at anytime, however, the change must not be.Bill of Rights Anchor Chart, 10 Amendments Anchor Chart, Social Studies Poster, U.S
Bill of Rights Anchor Chart 10 Amendments Anchor Chart Etsy
Constitutions and Contracts Amending or Changing the Contract United States Government
PPT The Constitution & Federalism PowerPoint Presentation, free download ID4690649
Periodic Table of Amendments Chart Teaching us history, Constitution activities, Civil war
List 97+ Pictures Pictures Of The Constitution Completed
First Amendment Anchor Chart, Bill of Rights Anchor Chart, Amendments Poster, United States
Fourth Amendment
Amendments South Carolina Public Charter School District
First Amendment Anchor Chart, Bill of Rights Anchor Chart, Amendments Poster, United States
Investigators With An Approved Protocol Must Submit An Amendment/Modification Of Research Form If There Are Significant Changes Involving Any Of The Study Protocols, Study Design,.
The Information Provided Here Builds On.
Related Post: