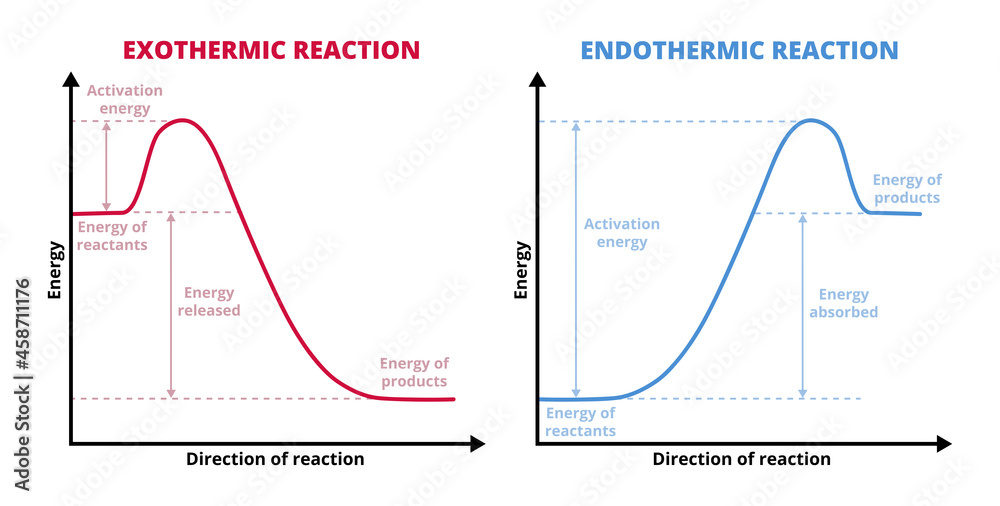
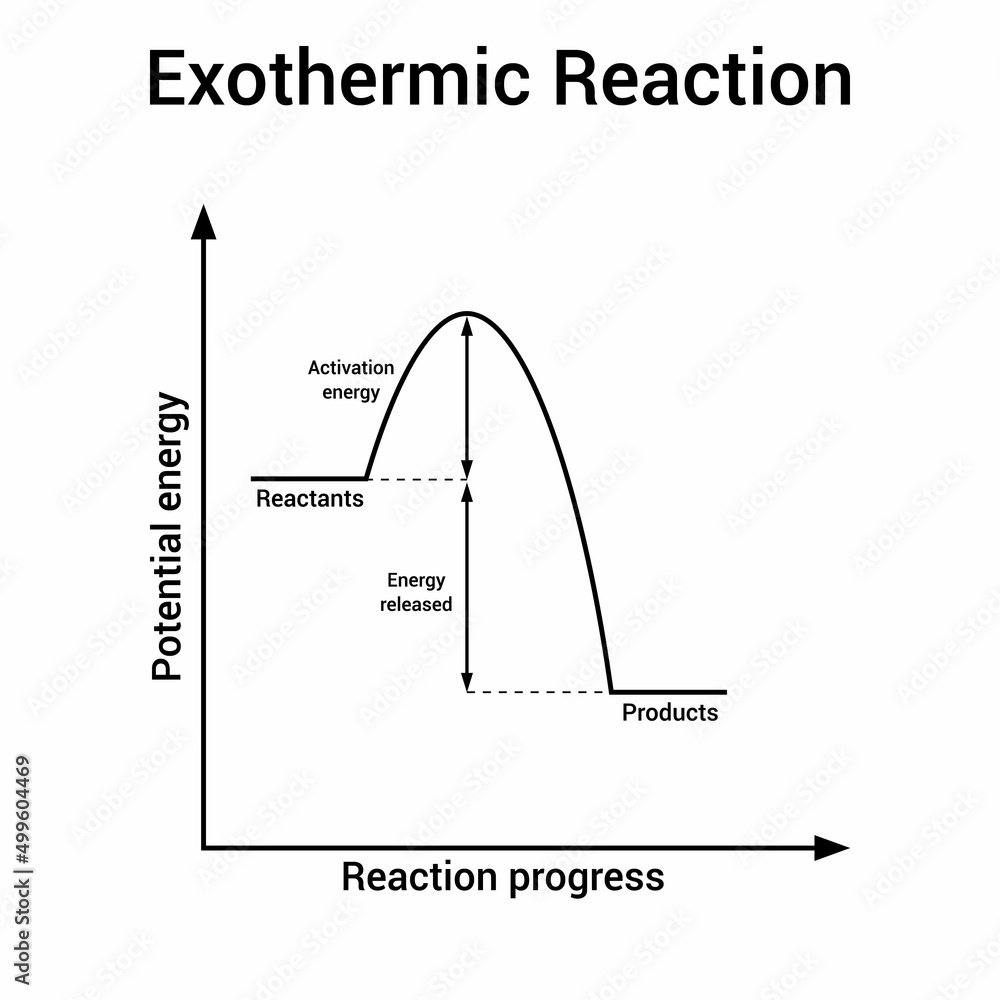
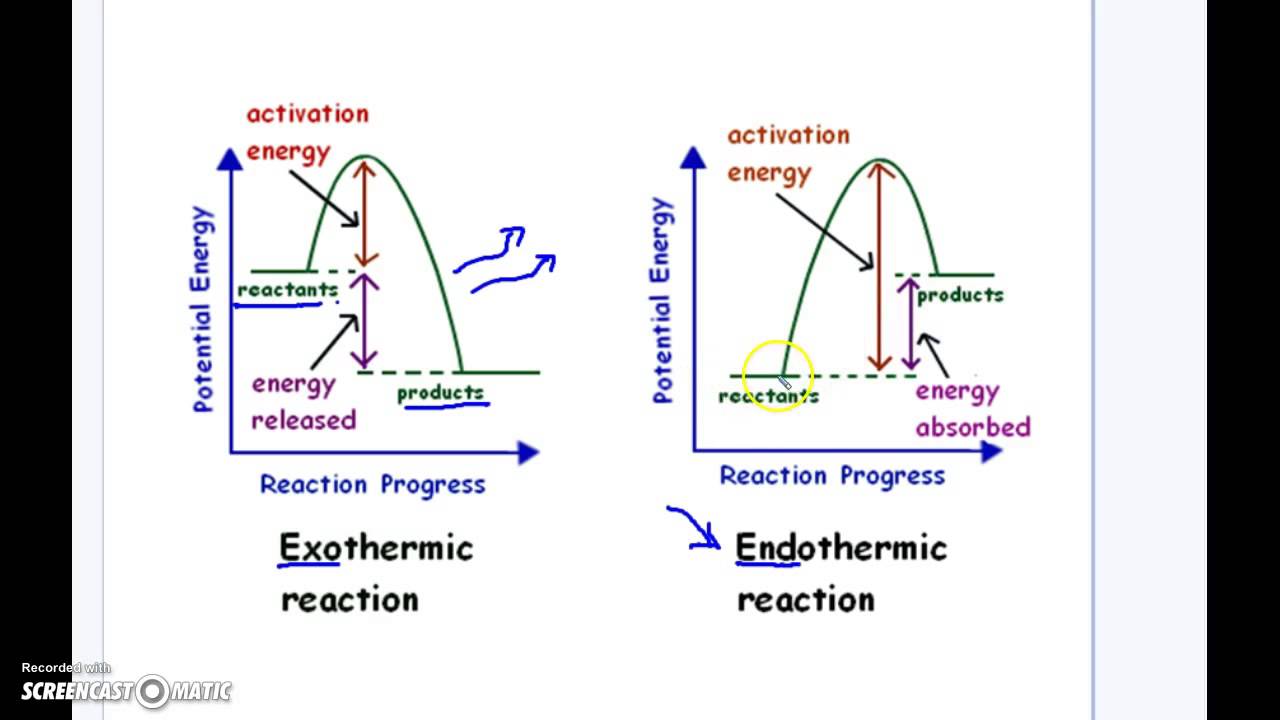
Exothermic Chart
Exothermic Chart - Examples include any combustion process, rusting of iron,. Exothermic reactions in the body (like atp hydrolysis) power muscles, nerves, and cellular machinery. The word itself is a compound word that supports this. Exothermic reaction in an exothermic reaction, energy is released to the surroundings. The system (reaction) releases heat to the surroundings as the reactants transform into products. An exothermic reaction is a type of chemical reaction that releases energy to its surroundings, primarily in the form of heat, but sometimes as light, electricity, or sound. A chemical reaction is said to be exothermic when it releases energy in the form of heat. Exothermic reactions these are reactions that transfer energy to the surroundings (ie the energy ex its from the reaction, hence the name ex othermic). A chemical reaction is exothermic if heat is released by the system into the surroundings. The reaction between hydrogen and oxygen is highly exothermic. The system (reaction) releases heat to the surroundings as the reactants transform into products. The prefix, ‘exo’ refers to ‘out’ or ‘outward’ (because heat energy is released. An exothermic reaction is a chemical reaction that gives off heat. Endothermic reactions are used for synthesis—building dna, proteins, and new cells. A chemical reaction is said to be exothermic when it releases energy in the form of heat. Exothermic reactions in the body (like atp hydrolysis) power muscles, nerves, and cellular machinery. Examples include any combustion process, rusting of iron,. An exothermic reaction is a type of chemical reaction that releases energy to its surroundings, primarily in the form of heat, but sometimes as light, electricity, or sound. In thermochemistry, an exothermic reaction is a reaction for which the overall standard enthalpy change δ h ⚬ is negative. [1][2] exothermic reactions usually release heat. An exothermic reaction is defined as a reaction that releases heat and has a net negative standard enthalpy change. An endothermic reaction occurs when energy is absorbed from the surroundings in the form of heat. An exothermic reaction is a type of chemical reaction that releases energy to its surroundings, primarily in the form of heat, but sometimes as light, electricity, or sound. Exothermic reactions these are reactions that transfer energy to the surroundings (ie the energy ex its. Exothermic reactions in the body (like atp hydrolysis) power muscles, nerves, and cellular machinery. An exothermic reaction is defined as a reaction that releases heat and has a net negative standard enthalpy change. The system (reaction) releases heat to the surroundings as the reactants transform into products. An endothermic reaction occurs when energy is absorbed from the surroundings in the. A chemical reaction is exothermic if heat is released by the system into the surroundings. The system (reaction) releases heat to the surroundings as the reactants transform into products. Exothermic reaction in an exothermic reaction, energy is released to the surroundings. Because the surroundings is gaining heat from the system, the temperature of the surroundings increases. Condensation and freezing are. The prefix, ‘exo’ refers to ‘out’ or ‘outward’ (because heat energy is released. Exothermic reactions in the body (like atp hydrolysis) power muscles, nerves, and cellular machinery. A chemical reaction is exothermic if heat is released by the system into the surroundings. Exothermic reaction in an exothermic reaction, energy is released to the surroundings. An exothermic reaction is a type. The prefix, ‘exo’ refers to ‘out’ or ‘outward’ (because heat energy is released. Exothermic reactions in the body (like atp hydrolysis) power muscles, nerves, and cellular machinery. Condensation and freezing are exothermic. An endothermic reaction occurs when energy is absorbed from the surroundings in the form of heat. The reaction between hydrogen and oxygen is highly exothermic. The word itself is a compound word that supports this. An exothermic reaction is a type of chemical reaction that releases energy to its surroundings, primarily in the form of heat, but sometimes as light, electricity, or sound. Exothermic reactions these are reactions that transfer energy to the surroundings (ie the energy ex its from the reaction, hence the name. A chemical reaction is exothermic if heat is released by the system into the surroundings. The prefix, ‘exo’ refers to ‘out’ or ‘outward’ (because heat energy is released. Condensation and freezing are exothermic. Examples include any combustion process, rusting of iron,. An endothermic reaction occurs when energy is absorbed from the surroundings in the form of heat. In thermochemistry, an exothermic reaction is a reaction for which the overall standard enthalpy change δ h ⚬ is negative. [1][2] exothermic reactions usually release heat. Exothermic reactions in the body (like atp hydrolysis) power muscles, nerves, and cellular machinery. An exothermic reaction is a type of chemical reaction that releases energy to its surroundings, primarily in the form of. Exothermic reactions in the body (like atp hydrolysis) power muscles, nerves, and cellular machinery. Because the surroundings is gaining heat from the system, the temperature of the surroundings increases. An endothermic reaction occurs when energy is absorbed from the surroundings in the form of heat. Exothermic reaction in an exothermic reaction, energy is released to the surroundings. The word itself. The prefix, ‘exo’ refers to ‘out’ or ‘outward’ (because heat energy is released. An endothermic reaction occurs when energy is absorbed from the surroundings in the form of heat. An exothermic reaction is a chemical reaction that gives off heat. An exothermic reaction is defined as a reaction that releases heat and has a net negative standard enthalpy change. Because. The reaction between hydrogen and oxygen is highly exothermic. Conversely, an exothermic reaction is one in which energy is released from the system into the. An exothermic reaction is a type of chemical reaction that releases energy to its surroundings, primarily in the form of heat, but sometimes as light, electricity, or sound. A chemical reaction is said to be exothermic when it releases energy in the form of heat. Because the surroundings is gaining heat from the system, the temperature of the surroundings increases. An exothermic reaction is a chemical reaction that gives off heat. The word itself is a compound word that supports this. Exothermic reaction in an exothermic reaction, energy is released to the surroundings. A chemical reaction is exothermic if heat is released by the system into the surroundings. Endothermic reactions are used for synthesis—building dna, proteins, and new cells. The system (reaction) releases heat to the surroundings as the reactants transform into products. Examples include any combustion process, rusting of iron,. Exothermic reactions in the body (like atp hydrolysis) power muscles, nerves, and cellular machinery. In thermochemistry, an exothermic reaction is a reaction for which the overall standard enthalpy change δ h ⚬ is negative. [1][2] exothermic reactions usually release heat. Condensation and freezing are exothermic.Vecteur Stock Vector graphs or charts of endothermic and exothermic reactions physics
Exothermic Reaction Coordinate Diagram Solved Reaction Consi
Exothermic Reaction Energy Diagram Exothermic Reaction Diagr
Exothermic And Endothermic Diagrams
PPT Unit 11 Thermochemistry PowerPoint Presentation, free download ID2132720
Vector graphs or charts of endothermic and exothermic reactions isolated on white. Exo and endo
Endothermic and Exothermic Reactions Lab ⋆ Exothermic reaction, Teaching
exothermic reaction graph in chemistry Stock Vector Adobe Stock
Exothermic And Endothermic Diagrams
Labelled Diagram Of Exothermic Reaction
The Prefix, ‘Exo’ Refers To ‘Out’ Or ‘Outward’ (Because Heat Energy Is Released.
Exothermic Reactions These Are Reactions That Transfer Energy To The Surroundings (Ie The Energy Ex Its From The Reaction, Hence The Name Ex Othermic).
An Endothermic Reaction Occurs When Energy Is Absorbed From The Surroundings In The Form Of Heat.
An Exothermic Reaction Is Defined As A Reaction That Releases Heat And Has A Net Negative Standard Enthalpy Change.
Related Post:
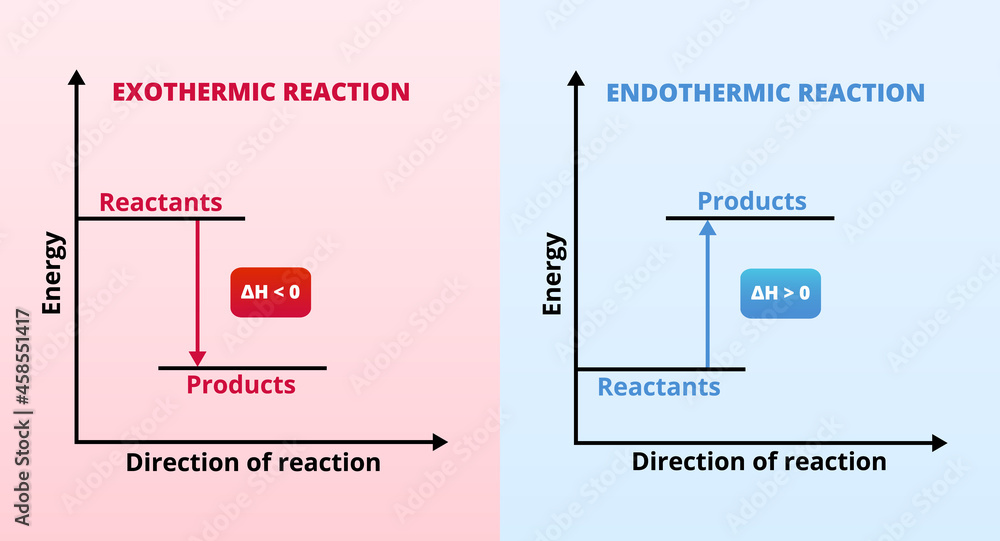


:max_bytes(150000):strip_icc()/endothermic-and-exothermic-reactions-602105_final-c4fdc462eb654ed09b542da86fd447e2.png)