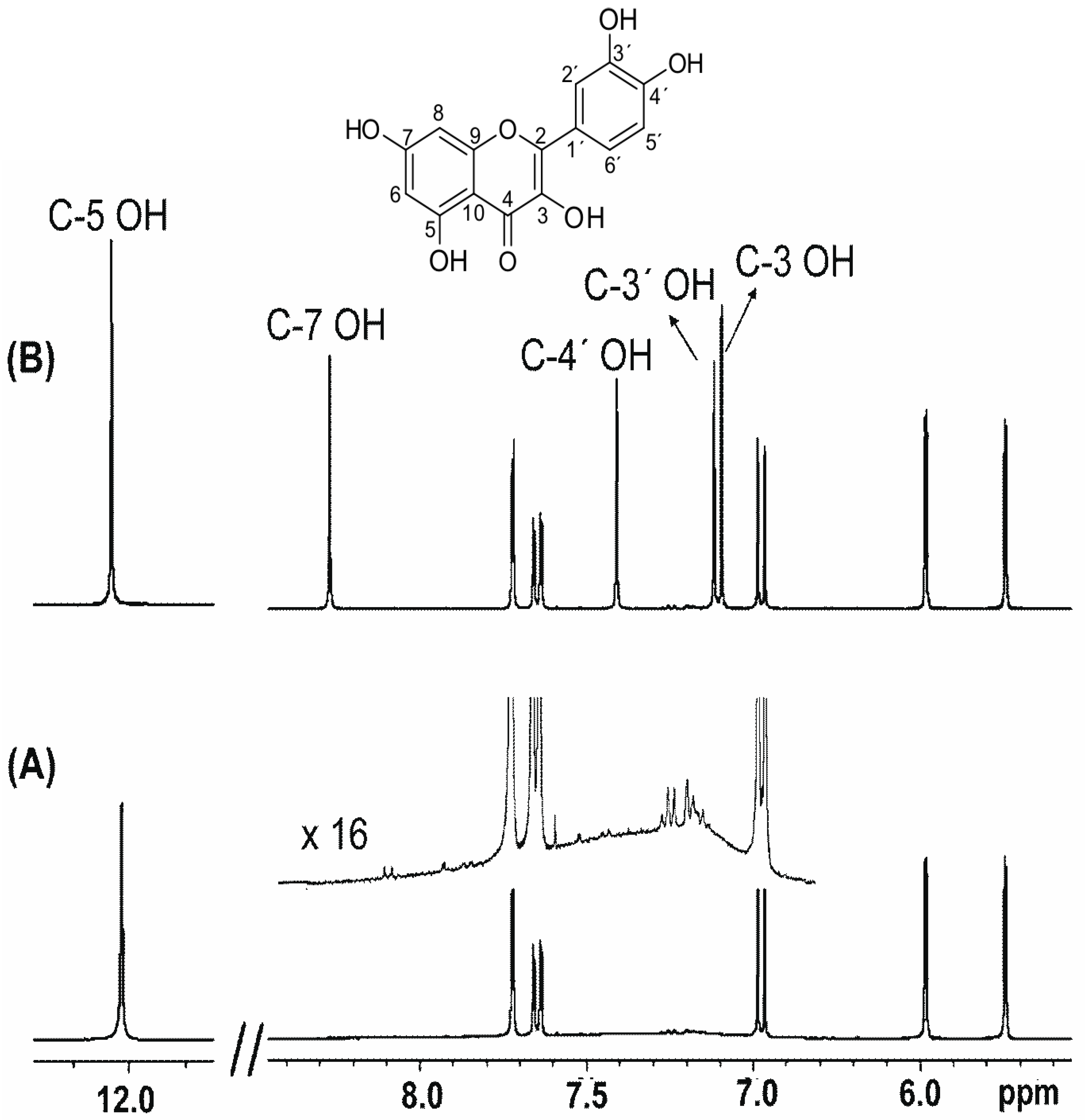
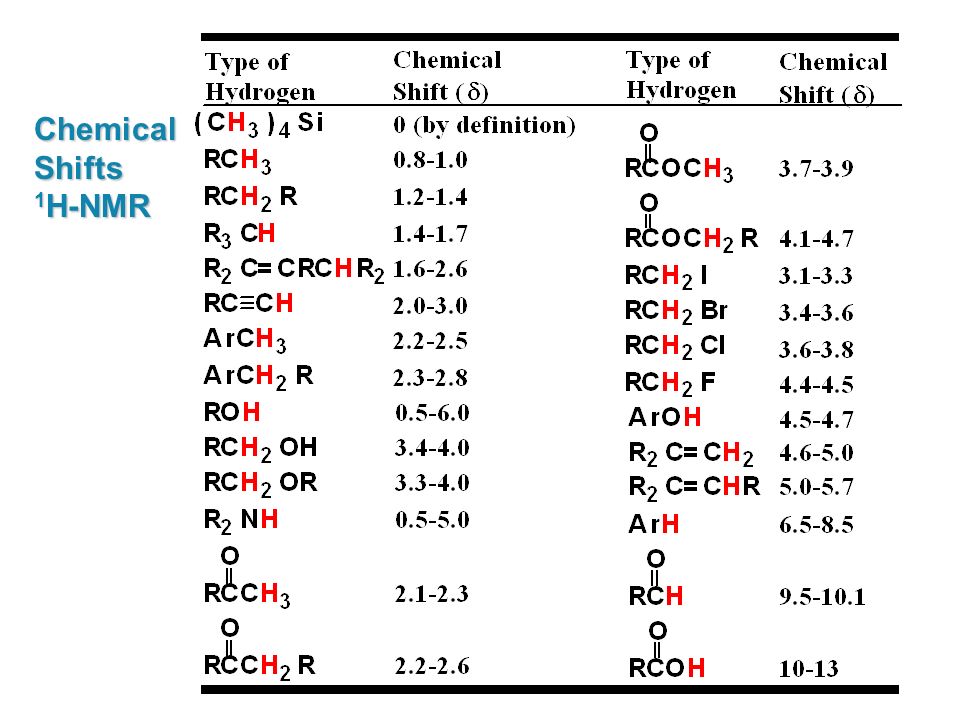
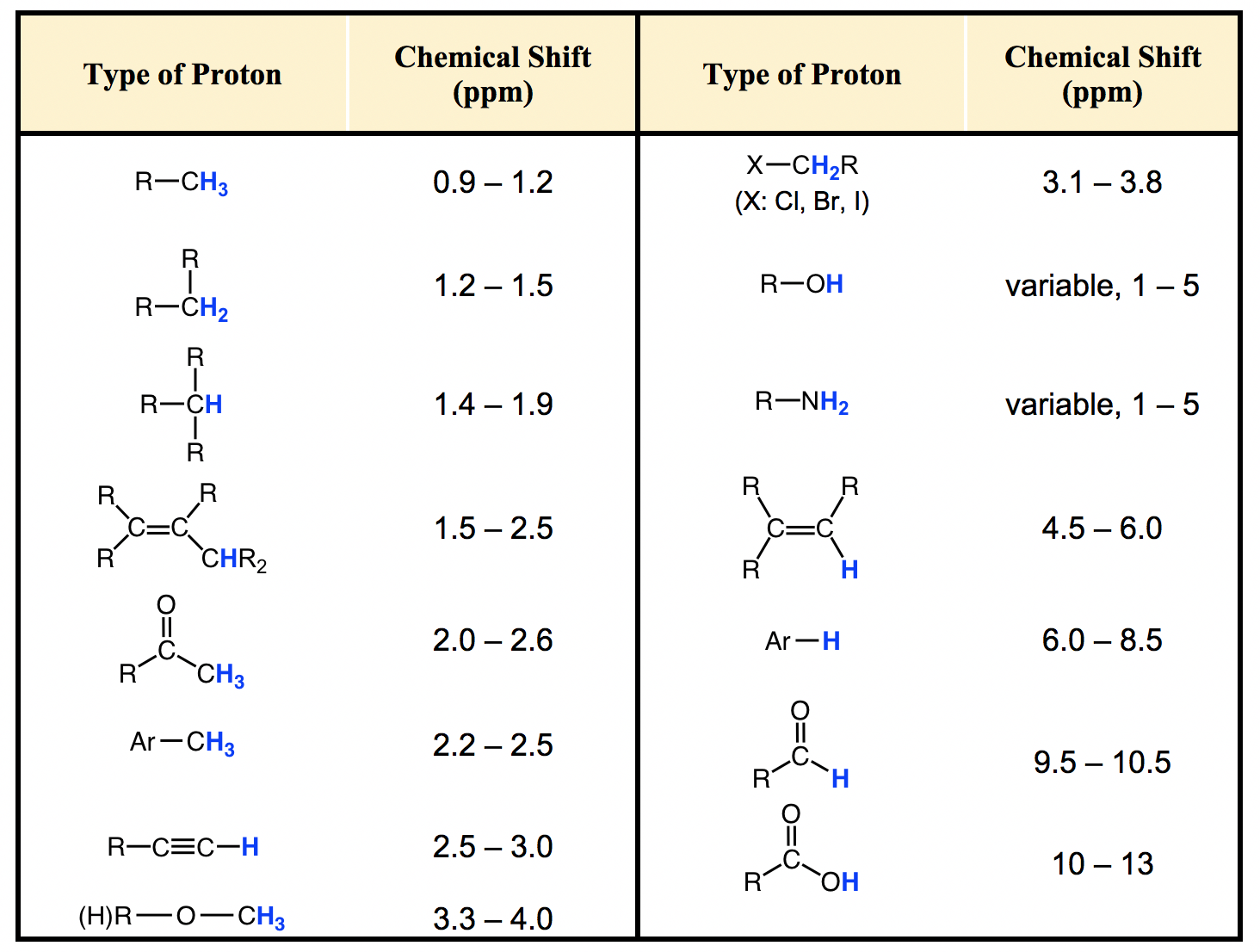
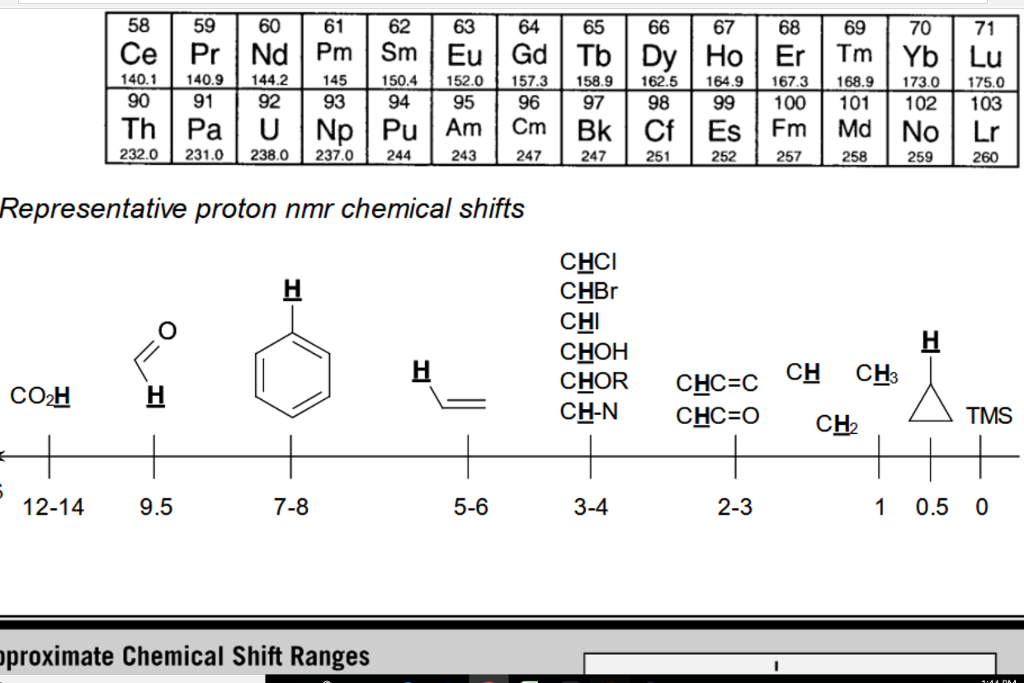
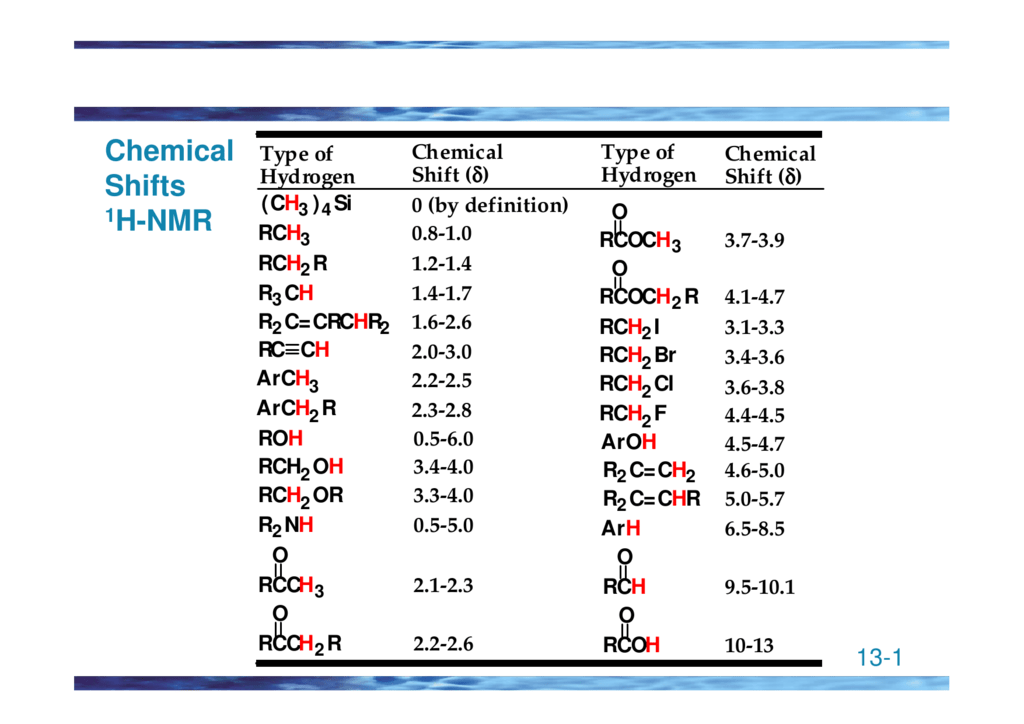
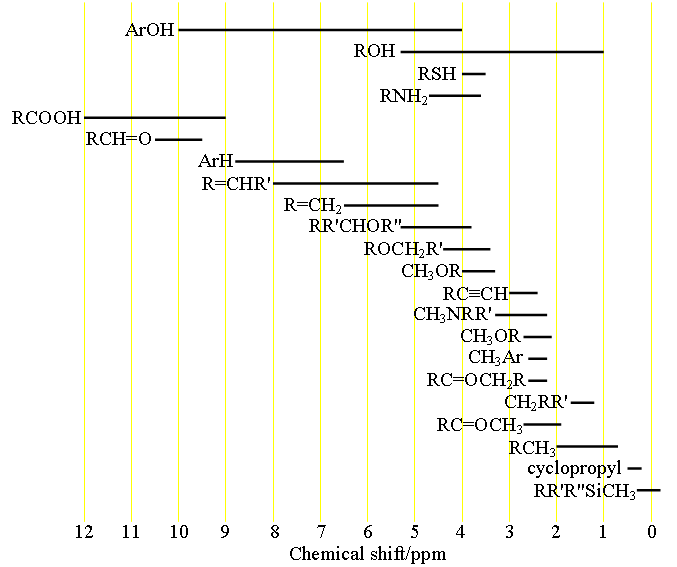
H Nmr Chart
H Nmr Chart - Nmr chemical shift and ppm value chart. Here we present the nmr shifts of the most commonly used solvents and impurities in organic synthesis measured in the 7 most frequently used deuterated solvents. From table 14.4 (labbook) or table h.6 (spec book) substituted alkanes 1. Overview of typical 1h nmr shifts. Alkene region modified from earlier handout It describes nuclear magnetic resonance (nmr) in details relevant to organic chemistry. Overview of typical 1h nmr shifts note: Table of characteristic proton nmr shifts. If a protic deuterated solvent is used (e.g., d2o or cd3od), then the nh and oh protons will exchange with the deuterium and the peaks will shrink or disappear entirely, since d (2h) does. In the nmr spectrum of the dianion, the innermost methylene protons (red) give an nmr signal at +22.2 ppm, the adjacent methylene protons (blue) give a signal at +12.6 ppm, and the methyl. Nmr chemical shift and ppm value chart. Here we present the nmr shifts of the most commonly used solvents and impurities in organic synthesis measured in the 7 most frequently used deuterated solvents. A guide to 1h nmr chemical shift values nuclear magnetic resonance (nmr) is a commonly used technique for organic compound structure determination. In the nmr spectrum of the dianion, the innermost methylene protons (red) give an nmr signal at +22.2 ppm, the adjacent methylene protons (blue) give a signal at +12.6 ppm, and the methyl. Overview of typical 1h nmr shifts note: Understanding the basics of nmr theory gets us ready to move on to the most important and practical part in this section, that is how to understand the 1h nmr spectrum and elucidate the. If a protic deuterated solvent is used (e.g., d2o or cd3od), then the nh and oh protons will exchange with the deuterium and the peaks will shrink or disappear entirely, since d (2h) does. Table of characteristic proton nmr shifts. You can download this chart as a printable acrobat pdf file. It describes nuclear magnetic resonance (nmr) in details relevant to organic chemistry. You can download this chart as a printable acrobat pdf file. Understanding the basics of nmr theory gets us ready to move on to the most important and practical part in this section, that is how to understand the 1h nmr spectrum and elucidate the. The effect of electronegativity and magnetic anisotropy on protons in upfield and downfield regions. If. Nmr chemical shift and ppm value chart. A guide to 1h nmr chemical shift values nuclear magnetic resonance (nmr) is a commonly used technique for organic compound structure determination. It also includes nmr summary data on coupling constants and chemical shift of. Understanding the basics of nmr theory gets us ready to move on to the most important and practical. Understanding the basics of nmr theory gets us ready to move on to the most important and practical part in this section, that is how to understand the 1h nmr spectrum and elucidate the. From table 14.4 (labbook) or table h.6 (spec book) substituted alkanes 1. The effect of electronegativity and magnetic anisotropy on protons in upfield and downfield regions.. A guide to 1h nmr chemical shift values nuclear magnetic resonance (nmr) is a commonly used technique for organic compound structure determination. If a protic deuterated solvent is used (e.g., d2o or cd3od), then the nh and oh protons will exchange with the deuterium and the peaks will shrink or disappear entirely, since d (2h) does. It describes nuclear magnetic. If a protic deuterated solvent is used (e.g., d2o or cd3od), then the nh and oh protons will exchange with the deuterium and the peaks will shrink or disappear entirely, since d (2h) does. It also includes nmr summary data on coupling constants and chemical shift of. A guide to 1h nmr chemical shift values nuclear magnetic resonance (nmr) is. You can download this chart as a printable acrobat pdf file. In the nmr spectrum of the dianion, the innermost methylene protons (red) give an nmr signal at +22.2 ppm, the adjacent methylene protons (blue) give a signal at +12.6 ppm, and the methyl. It describes nuclear magnetic resonance (nmr) in details relevant to organic chemistry. Understanding the basics of. A guide to 1h nmr chemical shift values nuclear magnetic resonance (nmr) is a commonly used technique for organic compound structure determination. Overview of typical 1h nmr shifts. Table of characteristic proton nmr shifts. It also includes nmr summary data on coupling constants and chemical shift of. It describes nuclear magnetic resonance (nmr) in details relevant to organic chemistry. You can download this chart as a printable acrobat pdf file. Overview of typical 1h nmr shifts note: If a protic deuterated solvent is used (e.g., d2o or cd3od), then the nh and oh protons will exchange with the deuterium and the peaks will shrink or disappear entirely, since d (2h) does. Here we present the nmr shifts of the. The effect of electronegativity and magnetic anisotropy on protons in upfield and downfield regions. Overview of typical 1h nmr shifts note: You can download this chart as a printable acrobat pdf file. Table of characteristic proton nmr shifts. In the nmr spectrum of the dianion, the innermost methylene protons (red) give an nmr signal at +22.2 ppm, the adjacent methylene. Table of characteristic proton nmr shifts. From table 14.4 (labbook) or table h.6 (spec book) substituted alkanes 1. Understanding the basics of nmr theory gets us ready to move on to the most important and practical part in this section, that is how to understand the 1h nmr spectrum and elucidate the. Nmr chemical shift and ppm value chart. The. Nmr chemical shift and ppm value chart. In the nmr spectrum of the dianion, the innermost methylene protons (red) give an nmr signal at +22.2 ppm, the adjacent methylene protons (blue) give a signal at +12.6 ppm, and the methyl. Here we present the nmr shifts of the most commonly used solvents and impurities in organic synthesis measured in the 7 most frequently used deuterated solvents. The effect of electronegativity and magnetic anisotropy on protons in upfield and downfield regions. A guide to 1h nmr chemical shift values nuclear magnetic resonance (nmr) is a commonly used technique for organic compound structure determination. It describes nuclear magnetic resonance (nmr) in details relevant to organic chemistry. Table of characteristic proton nmr shifts. Understanding the basics of nmr theory gets us ready to move on to the most important and practical part in this section, that is how to understand the 1h nmr spectrum and elucidate the. Alkene region modified from earlier handout From table 14.4 (labbook) or table h.6 (spec book) substituted alkanes 1. If a protic deuterated solvent is used (e.g., d2o or cd3od), then the nh and oh protons will exchange with the deuterium and the peaks will shrink or disappear entirely, since d (2h) does. You can download this chart as a printable acrobat pdf file.H Nmr Spectrum Chart
H Nmr Chemical Shift Chart Ponasa
6.6 ¹H NMR Spectra and Interpretation (Part I) Organic Chemistry I
NMR Spectroscopy Principles, Interpreting an NMR Spectrum and Common Problems Technology Networks
H Nmr Chemical Shift Chart Ponasa
H Nmr Spectroscopy Table at Lois Coffman blog
Analytical Chemistry A Guide to Proton Nuclear Resonance (NMR) Compound Interest
Nmr Shift Chart vrogue.co
H Nmr Chemical Shift Chart Ponasa
Nmr Values Chart
Overview Of Typical 1H Nmr Shifts Note:
It Also Includes Nmr Summary Data On Coupling Constants And Chemical Shift Of.
Overview Of Typical 1H Nmr Shifts.
Related Post: